|
It suggests that there is more involved than simply overcoming a larger ionic charge. The third IE, however, is over five times the previous one. The second IE is twice the first, which is not a surprise: the first IE involves removing an electron from a neutral atom, while the second one involves removing an electron from a positive ion. Mg(g) \longrightarrow Mg (g) e − IE 1 = 738 kJ/mol For example, the following are the first three IEs for Mg, whose electron configuration is 1 s 22 s 22 p 63 s 2: However, IE takes a large jump when a successive ionization goes down into a new shell. This is because more than one IE can be defined by removing successive electrons (if the atom has them to begin with):Ī(g) \longrightarrow A (g) e − IE 1Ī (g) \longrightarrow A 2 (g) e − IE 2Ī 2 (g) \longrightarrow A 3 (g) e − IE 3Įach successive IE is larger than the previous because an electron is being removed from an atom with a progressively larger positive charge. IE also shows an interesting trend within a given atom. Again, the trend isn’t absolute, but the general trends going across and down the periodic table should be obvious.įigure 2. However, as you go across the periodic table and the electrons get drawn closer in, it takes more energy to remove an electron as a result, IE increases.įigure 2 “Ionization Energy on the Periodic Table” shows values of IE versus position on the periodic table. As you go down the periodic table, it becomes easier to remove an electron from an atom (i.e., IE decreases) because the valence electron is farther away from the nucleus. It is always positive because the removal of an electron always requires that energy be put in (i.e., it is endothermic). IE is usually expressed in kJ/mol of atoms. Ionization energy (IE) is the amount of energy required to remove an electron from an atom in the gas phase:Ī(g) \longrightarrow A (g) e − ΔH ≡ IE 
We use the periodic table and our knowledge of the trends in atomic size further up and to the right are the smaller atoms. Figure 1 “Atomic Radii Trends on the Periodic Table” shows spheres representing the atoms of the s and p blocks from the periodic table to scale, showing the two trends for the atomic radius.įor the following elements, write them in order of smallest to largest, using only the periodic table: Atoms get smaller as you go from left to right across a period, and get larger as you go down a group. The size of atoms vary and there are two periodic trends. Such radii can be estimated from various experimental techniques, such as the x-ray crystallography of crystals. Although the concept of a definite radius of an atom is a bit fuzzy, atoms behave as if they have a certain radius. The atomic radius is an indication of the size of an atom. There may be a few points where an opposite trend is seen, but there is an overall trend when considered across a whole row or down a whole column of the periodic table. 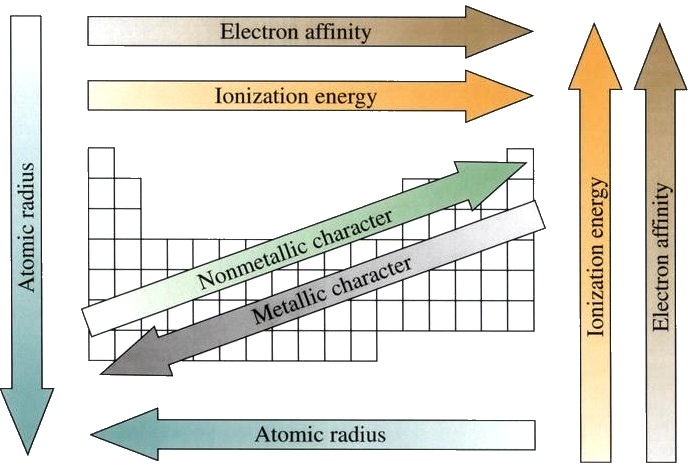
There is no other tool in science that allows us to judge relative properties of a class of objects like this, which makes the periodic table a very useful tool. 
The variation of properties versus position on the periodic table is called periodic trends. One of the reasons the periodic table is so useful is because its structure allows us to qualitatively determine how some properties of the elements vary versus their position on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
